Reaping 2020’s Nobel prize in chemistry, Emmanuelle Charpentier and Jennifer A. Doudna have made a landmark discovery for the CRISPR/Cas 9 method employed in genome editing. Clustered, regularly interspaced short palindromic repeats associated proteins (CRISPR/Cas 9) are used to edit segments of the genome (complete genetic makeup of an organism) thus modifying an organism’s genome by adding, deleting, or substituting the desired segment of it. CRISPR/Cas 9’s development and application is receiving the limelight as it is claimed to be efficient, faster, and accurate among all gene-editing tools.
CRISPR was first discovered in bacteria and was considered as acquiring immunity to combat infections caused by bacteriophages (viruses that cause infect bacterial cells). When a bacteriophage inserts its DNA into the host bacterial cell, a short sequence is inserted between the CRISPR sequence region. CRISPR region has spaces between them that store memory of each infection that the bacterial cell was exposed to. In case of repeated exposure to the same infection, bacterial cells can then fight back to the infection.
The recent discovery reels the roots of the time when Emmanuelle was working on the bacterium Streptococcus pyogenes and came across tracr RNA. Her research proved tracr RNA as a part of bacteria’s ancient immune system: CRISPR/Cas9. She published her findings in 2011. During that time, Emmanuelle collaborated with Jennifer A. Doudna who was equipped with vast RNA knowledge.
Together, both the women altered the genetic scissors and proved that they could be modified for the desired use.
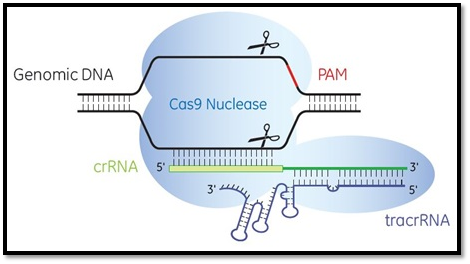
The work of Emmanuelle and Jennifer illustrated the complete working mechanism of the CRISPR/Cas9 molecule. CRISPR/Cas 9 comprises two prime molecules: Cas 9 (enzyme) and RNA. Reflecting on Cas 9, it functions like molecular scissors that cut DNA at specific sites or the gene of interest. The segment that needs to be removed, replaced, or added at a specific location is isolated using Cas 9.
The second molecule is the piece of RNA also referred to as the guide RNA. It is a short sequence of pre-formed bases located within a larger vicinity of the RNA scaffold. The larger RNA scaffold translocates and binds to DNA, after which the pre-formed base sequence guides the Cas9 enzyme to attack the target site (the part of DNA that needs to be spliced). As the guide RNA has a sequence of bases complementary to the target sequence on the DNA strand, the efficiency of the procedure is enhanced as the likelihood of guide RNA binding to a non-targeted site on DNA is highly minimized preventing any errors.
Once guide RNA tracks the pathway for Cas9 at a target site, Cas 9 then cuts the target gene sequence by splicing the segment from both ends on the DNA strand. At this stage, the cell detects a change in its DNA and initiates a mechanism to counter this change by using its DNA repair mechanisms. Cell’s DNA repair machinery can be exploited and modified to introduce changes in the genome.
The significance of this discovery lies in the fact that it can be used in numerous areas of biochemistry and biotechnology to treat genetic diseases, enhance the productivity of genetic engineering, and derive effective ways to tackle infections as mentioned below:
Agricultural advancements
Acknowledged as an accelerated form of selective breeding by United States Department of Agriculture, CRISPR/Cas9 can be maneuvered to attain high quality and safer food products. Nutrient food can be boosted, disease-resistant plants can be bred, and drought-resistant plants can be produced.
For instance, the wheat plant’s genome contains an MLO gene that makes the plant prone to powdery mildew. CRISPR/Cas9 can be manipulated to remove the MLO gene from the whet genome thus producing normal plants. Similarly, in the soybean plant, many of its traits, such as herbicide resistance and enhanced protein composition, are introduced, and their expression has been enhanced using CRISPR/Cas9 scissors.
Moreover, allergy-free foods can be produced using this technology so individuals who are allergic to food components can then consume that without risking their health. Work is underway on many plant seeds such as maize, rice, and tomato to modify their consumer demands and increase the yield.
Greener Fuels
CRISPR/Cas9 can harness biofuels by using algae. “Synthetic genomics,” a company that has initiated procedures of producing such algae strains that yield twice the amount of fats. Genes that decrease fat production are spliced out using this technology. Obtained fats are then manipulated to produce biofuel.
Genetic Diseases
Cystic fibrosis is a genetic disease caused by a mutation in the CFTR gene (Cystic fibrosis transmembrane conductance regulator). THE defective CFTR gene hinders the passage of water and ions in and out of cells, resulting in the accumulation of thick secretions like mucus. These secretions clog the airway and support bacterial growth. Gene editing using CRISPR/Cas9 aims to cleave the CFTR gene’s mutated DNA sequence and replace it using the normal CFTR gene. The vector utilized in this procedure is the lipid nanoparticle (LNP) or Adeno-associated virus (AVV). Editas Medicine has undertaken this approach to treat Cystic Fibrosis and carry out human trials to see the effectiveness of this technology.

De-extinction
Scientists are applying CRISPR/Cas9 technology to introduce extinct animals’ genes into present day animals, producing hybrids. Those hybrids will then be interbred over generations until the offspring’s DNA matches the original, extinct animal’s DNA.
One might find this implausible, but this is no longer a daydream. George Church, a Harvard biologist and trailblazer of CRISPR, has devoted eleven years of his life to finding the mechanism to bring extinct animals back. His subject is an ancient mammoth, whose DNA samples can be obtained from its skin, muscle, and fur buried under the snow. Along with his team, he has reconstructed the DNA sequence code and read it, giving them an insight into the genome of mammoth that existed thousands of years ago. The basis of de-extinction rest is reading the extinct species’ DNA code and then manipulating the genome of its closely related species to prevent extinction. However, he and his team aim to create an elephant-mammoth hybrid using CRISPR/Cas 9 technology to save the Asian elephants on the verge of extinction.
Similarly, work is being done on Pigeon and Tiger to restore and prevent the extinction of the species left on the planet using improved genetic engineering techniques.
Animal and cell models
CRISPR/Cas9 has opened a new domain in scientific research. Its mechanism of action can aid in the production of in vivo and in vitro disease models. The most common method of producing animal models using genetic engineering is using fertilized stage 1 embryos. CRISPR/Cas9 and fertilized embryos are operated together to create models using techniques such as microinjection, electroporation, and genome editing via oviductal nucleic acid delivery (GONAD).
Microinjection enables entry of Cas9/g-RNA complex into the cytoplasm of a fertilized embryo. Electroporation allows gene editing using electric stimulation, and GONAD is employed to introduce Cas9/g-RNA into embryos without risking the life of subject animal. The mouse is the common subject for animal models and the techniques mentioned above are used to generate mouse models to study diseases.
The ability to manipulate the genome to generate animal models, use them to study diseases, and find a potential cure for diseases is one of the prime advantages this technology offers. This application’s significance is a great one as it can possibly and treatments to diseases like cancer and HIV.
The above-mentioned domains are a few of the many fields that can benefit from CRISPR/Cas9 molecular scissors. Nothing is impossible when it comes to science and research. There is more than one can envision and imagine. Emmanuelle Charpentier and Jennifer A. Doudna’s research of CRISPR/Cas technology might be the key to the lock of numerous unanswered questions.
“To raise new questions, new possibilities, to regard old problems from a new angle, requires creative imagination and mark a real advance in science” – Albert Einstein.
References
- https://www.ntfe.no/i/2019/2/tfe-2019-02b-1490
- https://www.yourgenome.org/facts/what-is-crispr-cas9
- https://www.cnet.com/features/using-crispr-to-resurrect-the-dead/
- https://www.tandfonline.com/doi/full/10.1080/19768354.2020.1726462
- https://cysticfibrosisnewstoday.com/crisprcas9-approach-for-cystic-fibrosis/

Maira Masood is a BS Biosciences student at NUST, Pakistan. She aspires to be a geneticist and wants to play an active part in spreading scientific awareness through writings.

